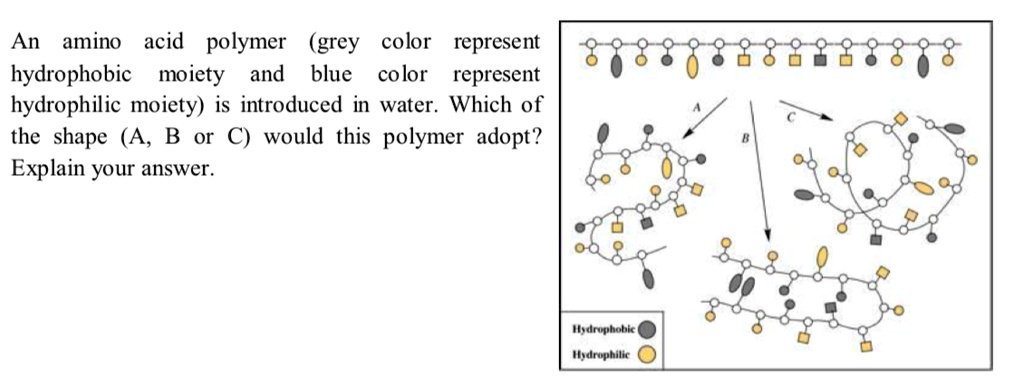
Hence, the hydrophobic nature of these compounds arises due to the side chains they have in their chemical structure. Since these amino acids are nonpolar, they cannot dissolve in water. Likewise, the name “hydrophobic” derives because it does not interact with water (“hydro” – water). Hydrophobic amino acids are a type of amino acids with a nonpolar nature. Summary What are Hydrophobic Amino Acids? Side by Side Comparison – Hydrophobic vs Hydrophilic Amino Acids in Tabular Formĥ. They are different from each other mainly based on the polarity. Moreover, we can categorize them as hydrophilic and hydrophobic amino acids, depending on their physicochemical nature. 
Furthermore, amino acids are mainly in two types as essential and nonessential amino acids. A protein is a giant polymer molecule which is an essential component of all living organisms. The key difference between hydrophobic and hydrophilic amino acids is that the hydrophobic amino acids are nonpolar whereas the hydrophilic amino acids are polar.Īmino acids are the building blocks of proteins.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
